What Is a Conjugate Acid Base Pair Apex
Unlike ions molecules carry no electrical charge. H 3 O OH -.

Union Celular Union Celular Ensenanza Biologia Biologia Celular
Why is water considered to be neutral.

. An acid base pair which differ from each other by a single proton H ion is called a conjugate pair. Tap card to see definition. On the other hand a conjugate base is merely what is left after an acid has donated a proton in a chemical reaction.
Acid Base HCl Cl- NH3 NH4 H2O H3O The acidbase numbering sytem. The concept of conjugate acid-base pair is related to Bronsted-Lowry acid-base theory and according to this theory acid is a proton H donor while base is a proton acceptor. HOCN and OCN - are an example of a conjugate acid-base pair.
Conjugate acid-base pair are compounds which differ by H Heres are two examples of conjugate acid-base pair. A pair of species in a reaction that differ by a proton. A Bronsted-Lowry base is a substance which accepts a proton or H ion from the other compound and forms conjugated acid.
All acids have a conjugate base and all bases have a conjugate acid. One of the PURINE or PYRIMIDINE components of NUCLEOTIDES. Similarly adding water to.
A more general definition is that a conjugate base is the base member X- of a pair of compounds that transform into each other by gaining or losing a proton. The acid that accepts an electron pair from a base C. What is a conjugate acid-base pair A conjugate acid is formed when a base gains a positive hydrogen Ion H and thus having the ability to lose this ion becomes a weak acid.
An acid and a base that are the same except for an H. Conjugate base is what is left over after an acid has donated a proton during a chemical reactionTherefore conjugate base is a species formed by the removal of a proton from an acid. A conjugate acid within the BrønstedLowry acidbase theory is a species formed by the reception of a proton H by a base in other words it is a base with a hydrogen ion added to it.
So when an acid reacts with base. The only difference between the two is a proton H. You know the precise concentration of the titrant.
The acid or base that you add to the unknown species in a titration. Apex Chemistry - 111 22 Terms. Two substances in a Brønsted-Lowry acid-base.
Two chemical different one H. Lets focus on the first example CH_3COOH. The species that is formed is the acids conjugate base.
The conjugate base of H3PO4 is H2PO4-. Conjugate acid base pair. Reaction whose chemical formulas differ by one.
There are two key terms associated with buffers. A Bronsted-Lowry acid is a substance which donates a proton or H ion to the other compound and forms a conjugated base. If you add an acid or a base to a buffered solution its pH will not change significantly.
What is a conjugate acid base pair. The conjugate acid of PO43- is HPO42-. An acid and a base that combine to form a molecule D.
When an acid dissociates into its ions in water it loses a hydrogen ion. According to the Brønsted-Lowry theory an acid is a proton donor and a base is a proton receptor. Ionic compounds such as common salt are made up not of molecules but of ions arranged in a crystalline structure.
The smallest particle of a compound that has all the chemical properties of that compound. An acid base pair which differ from each other by a single protonH ion is called a conjugate pair. The base dissolves in water with the production of hydroxyl ions and reacts with acids to form salts.
A chemical substance which has a tendency to accept protons H. See answers 2 Best Answer. What Is a Buffer.
Strong acids and bases ionize completely in an aqueous solution whereas weak acids and bases are partially ionized. The conjugate base is able to gain or absorb a proton in a chemical. Molecules are made up of two or more atoms either of the same element or of two or more different elements.
From the list of moleculeion pairs below click on those that are conjugate acid-base pairs. In the Bronsted-Lowry theory an acid is a proton donor and a base is a proton acceptor. Apex Chemistry - 113 34 Terms.
Having no missing parts. A buffer is an aqueous solution that has a highly stable pHA buffering agent is a weak acid or weak base that helps maintain the pH of an aqueous solution after adding another acid or base. Proton H Click again to see term.
An acid and a base that react together to form a salt B. What is a conjugate acid-base pair. Click card to see definition.
The partners in an acidbase conjugate pair differ from one other by a single hydrogen ion. To find out exactly who someone is or what something is. Is H3PO4 and PO4 3 a conjugate acid base pair.

Pdf Coupling Apex Labeling To Imaging Mass Spectrometry Of Single Organelles Reveals Heterogeneity In Lysosomal Protein Turnover
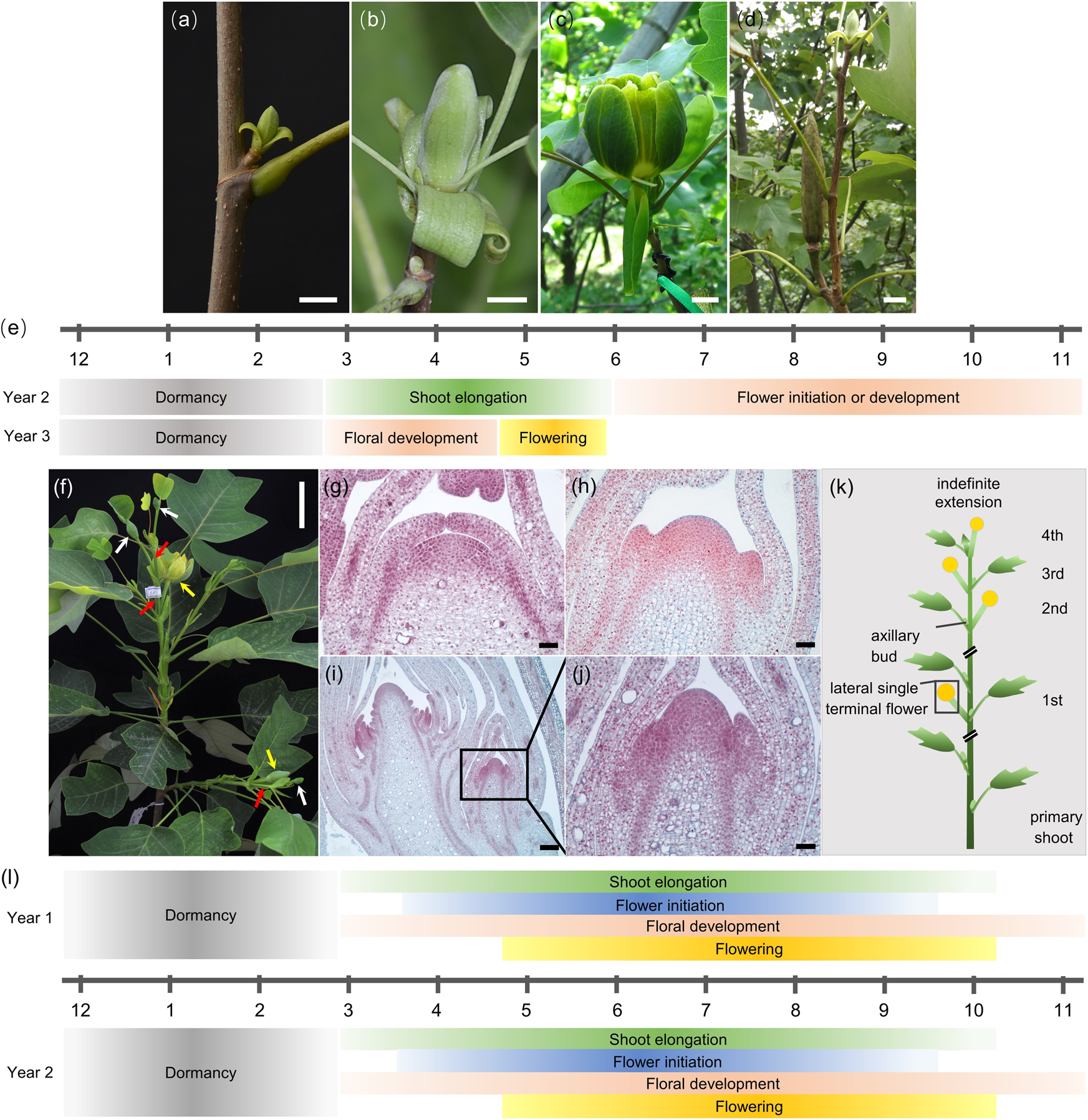
Morphological Phenological And Transcriptional Analyses Provide Insight Into The Diverse Flowering Traits Of A Mutant Of The Relic Woody Plant Liriodendron Chinense Horticulture Research

Pdf Apex Fingerprinting Reveals The Subcellular Localization Of Proteins Of Interest

Pdf Coupling Apex Labeling To Imaging Mass Spectrometry Of Single Organelles Reveals Heterogeneity In Lysosomal Protein Turnover

Pdf Apex Fingerprinting Reveals The Subcellular Localization Of Proteins Of Interest

Pdf Apex Fingerprinting Reveals The Subcellular Localization Of Proteins Of Interest

The Paperclip Triplex Understanding The Role Of Apex Residues In Tight Turns Biophysical Journal

Apex 8 2 Key Terms C Flashcards Quizlet

Pex18p Is Constitutively Degraded During Peroxisome Biogenesis Journal Of Biological Chemistry
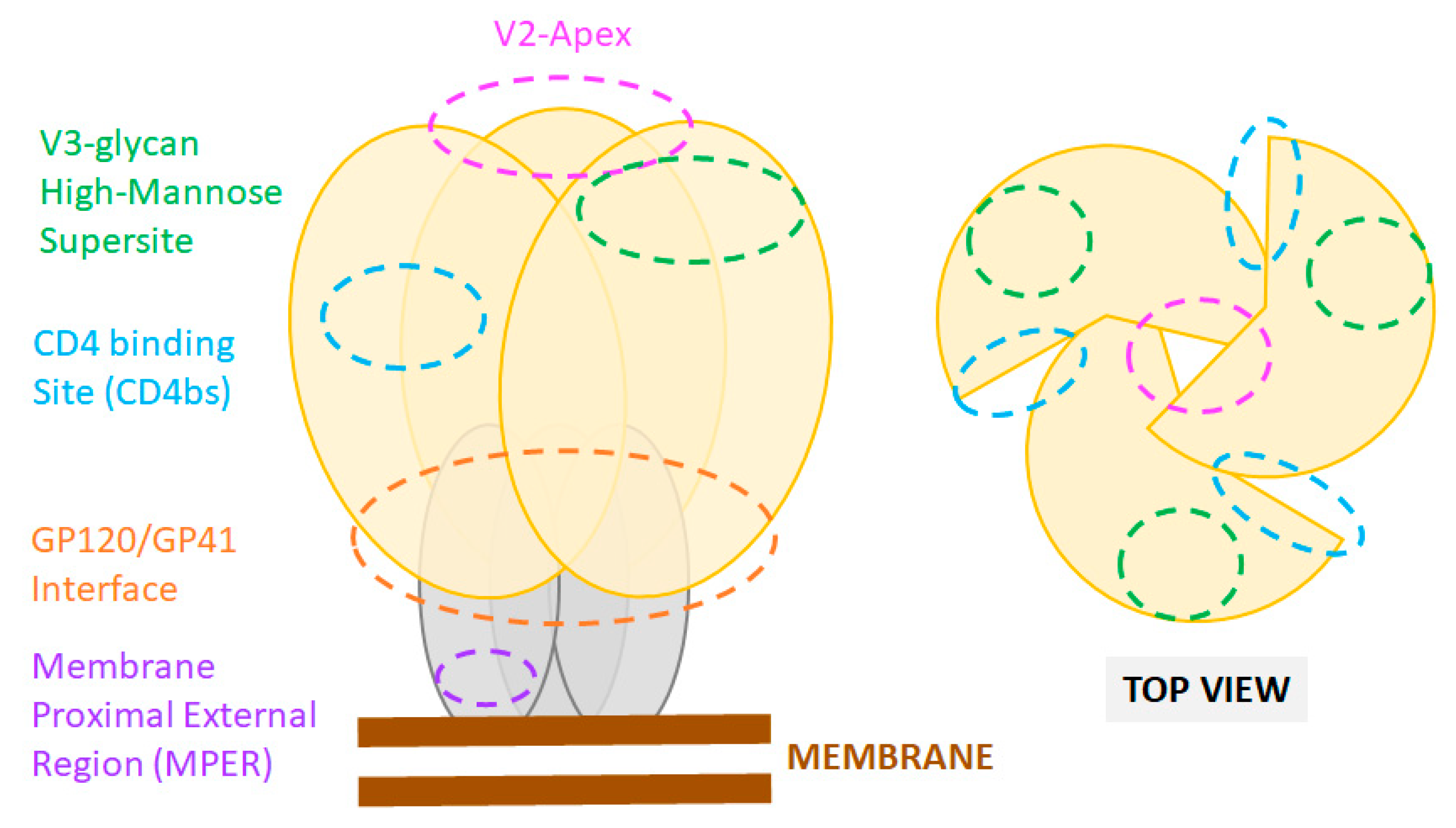
Microorganisms Free Full Text Hiv 1 Entry And Prospects For Protecting Against Infection Html

Pex18p Is Constitutively Degraded During Peroxisome Biogenesis Journal Of Biological Chemistry

Pdf Coupling Apex Labeling To Imaging Mass Spectrometry Of Single Organelles Reveals Heterogeneity In Lysosomal Protein Turnover

Horticulturae Free Full Text Insights Into Factors Controlling Adventitious Root Formation In Apples Html

Pdf Coupling Apex Labeling To Imaging Mass Spectrometry Of Single Organelles Reveals Heterogeneity In Lysosomal Protein Turnover

Union Celular Union Celular Ensenanza Biologia Biologia Celular

Directed Evolution Of Apex2 For Electron Microscopy And Proximity Labeling Abstract Europe Pmc



Comments
Post a Comment